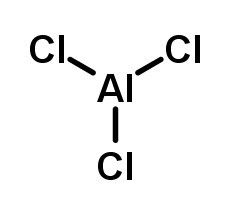
How to arrange the following according to increasing boiling point, freezing point, and vapour pressure: 1m NaCl, 1m CaCl2,1m C6H12O6, and 1m AlCl3 - Quora

The correct relationship between the boiling points of very dilute solutions of AlCl3(T1) and CaCl2(T2) , having the same molar concentration is:

Catalytic Dehydration of Carbohydrates Suspended in Organic Solvents Promoted by AlCl3/SiO2 Coated with Choline Chloride - Yang - 2015 - ChemSusChem - Wiley Online Library
Survey of Properties of Key Single and Mixture Halide Salts for Potential Application as High Temperature Heat Transfer Fluids f

Pressure-temperature phase diagram of AlCl 3 including (extrapolation... | Download Scientific Diagram
Molality Chart Problem Problem Convert Convert Colligative Properties Where it is used Boiling Water

The correct relationship between the boiling points of very dilute solutions of AlCl3(t1) and CaCl2(t2) , having the same molar concentration, is :