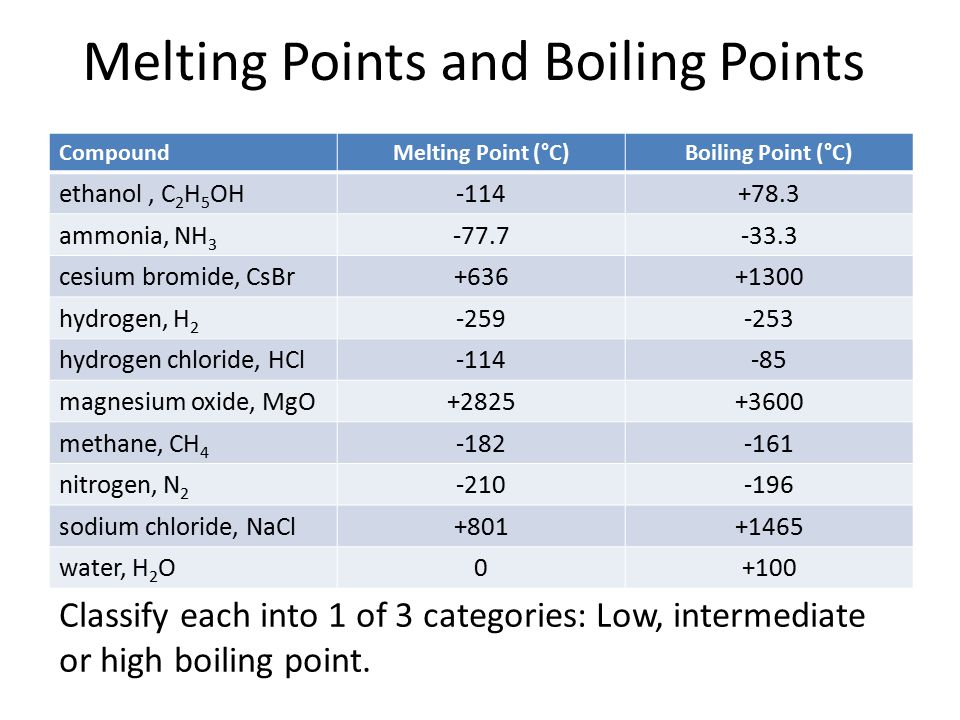
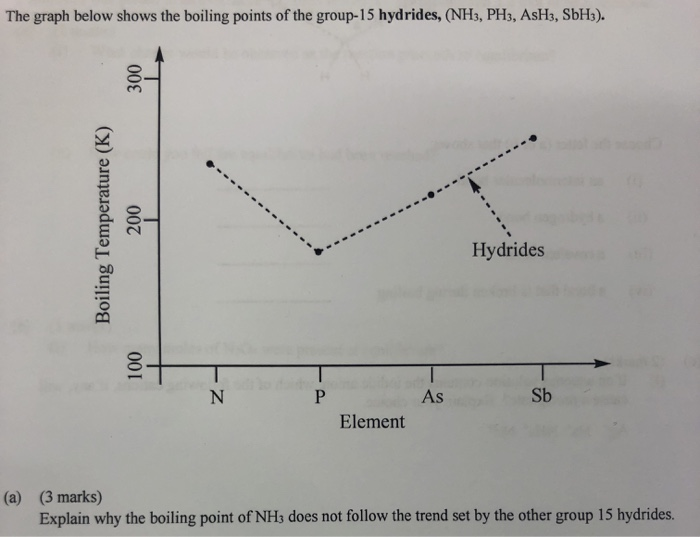
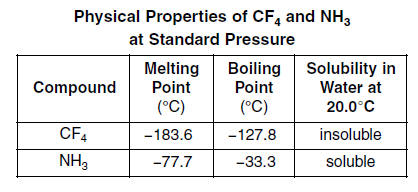
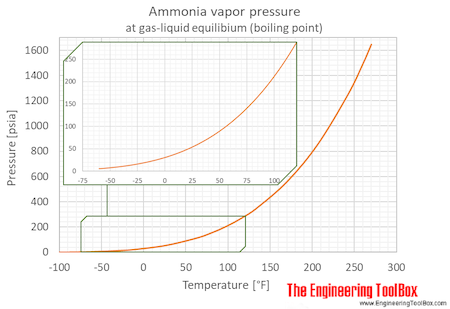
SOLVED: The normal boiling points of ammonia, water, and hydrogen fluoride increase in the order NHs (bp = -33 C) < HF (bp 19 C) HzO (bp = 100 oC): What is

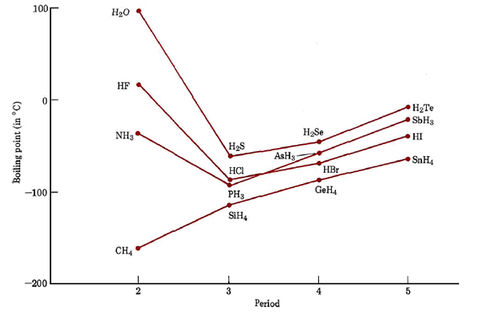
A diagram illustrating the "unexpected" rise in boiling point (Y-axis)... | Download Scientific Diagram

The boiling point of phosphine, PH3 (-88 degrees C) is lower than that of ammonia, NH3 (-33 degrees C) even though phosphine has twice the molar mass of NH3. Why? | Homework.Study.com