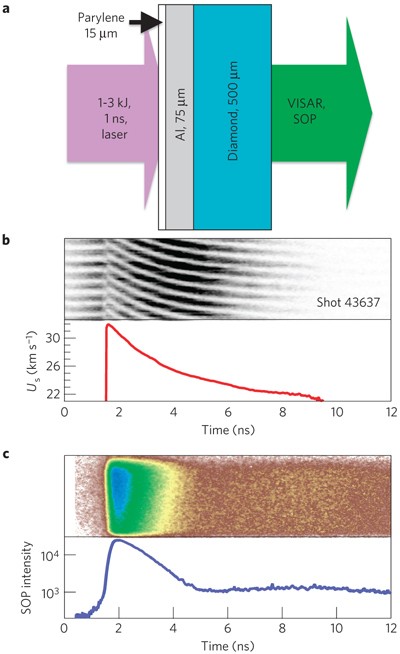
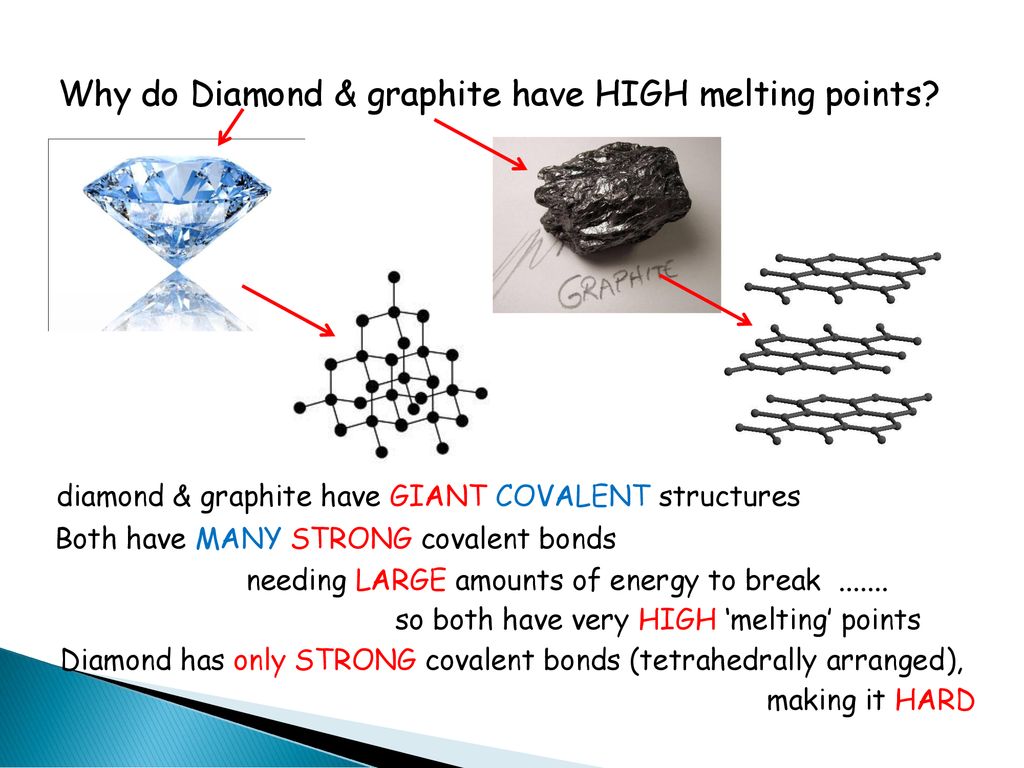
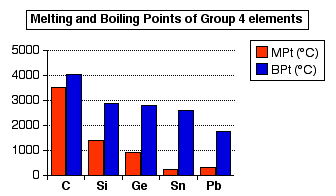
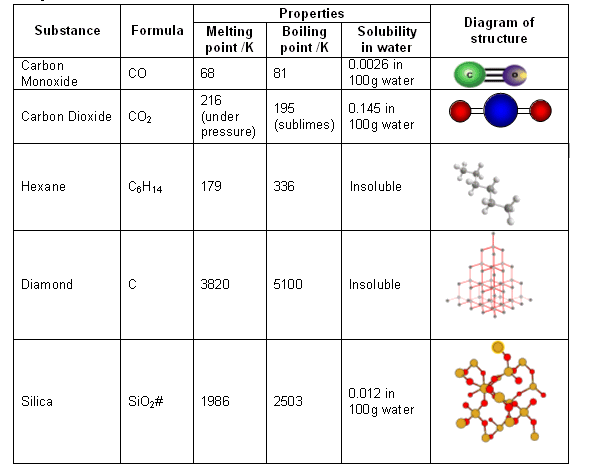
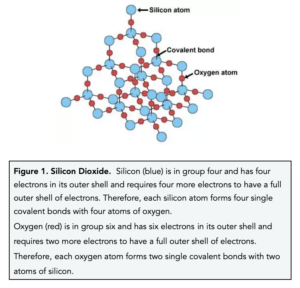
How hard is it to melt diamond? Can I make profit melting many little diamonds into one big diamond? I've heard scientists have melted diamond at high temperature and pressures with strong
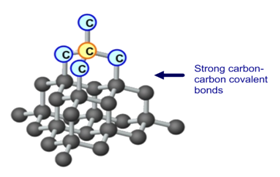
1:49 explain why substances with giant covalent structures are solids with high melting and boiling points - TutorMyself Chemistry
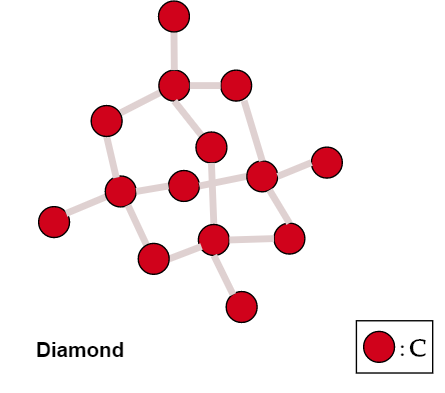
The high melting and boiling points of diamond are due to:A. Its high refractive indexB. Its high IP valueC. Giant polymer structure with strong covalent bondsD. Its high electronegativity