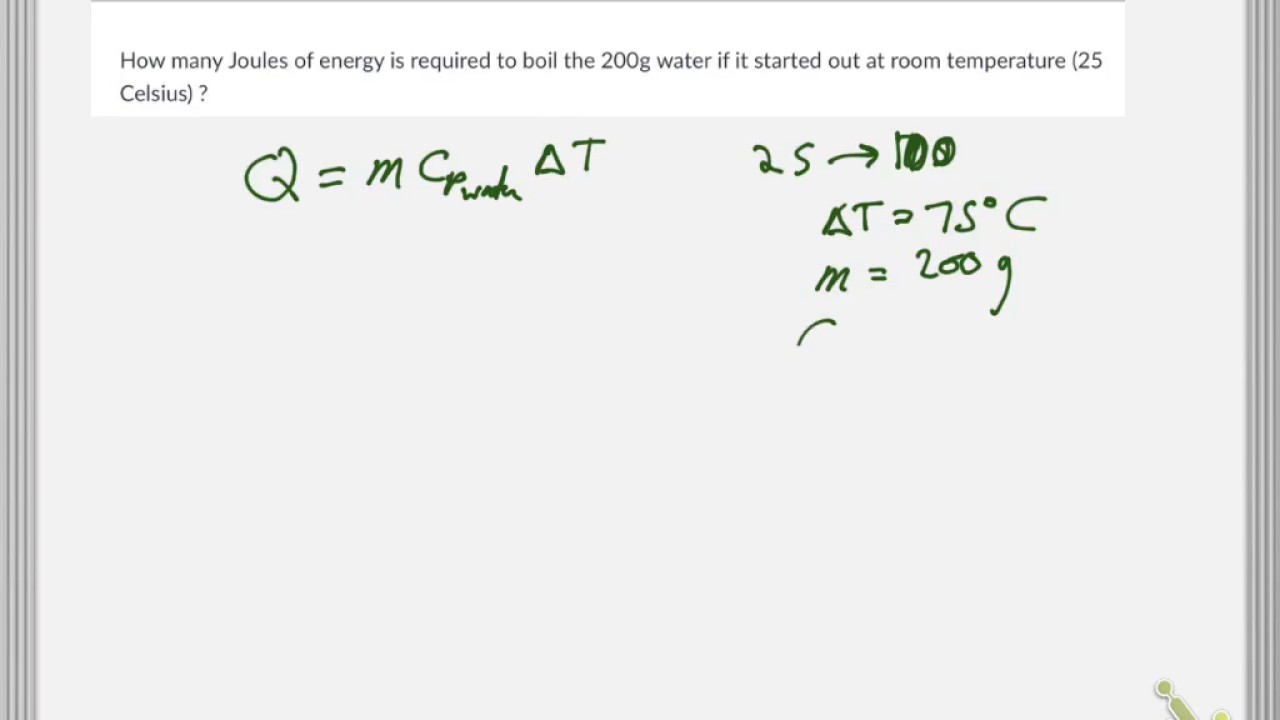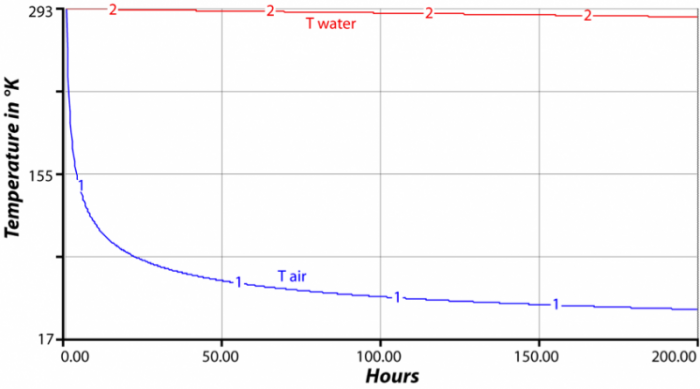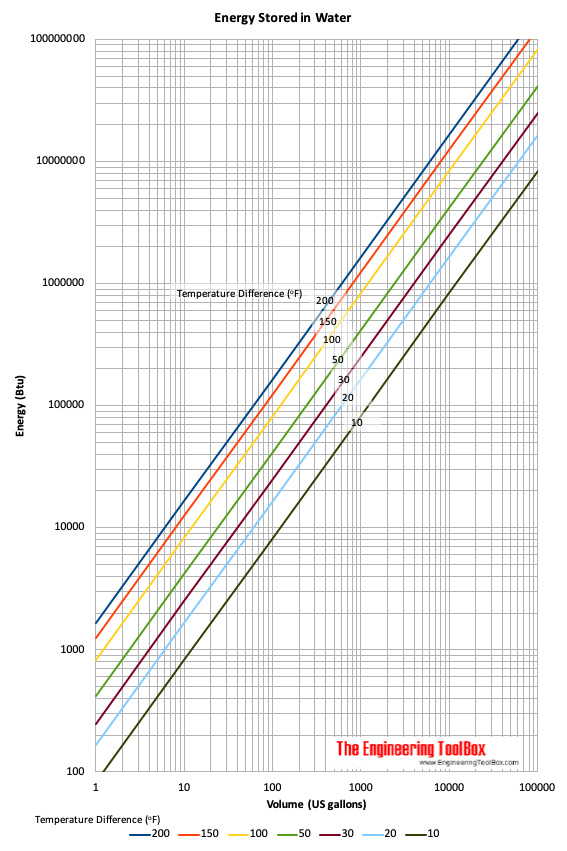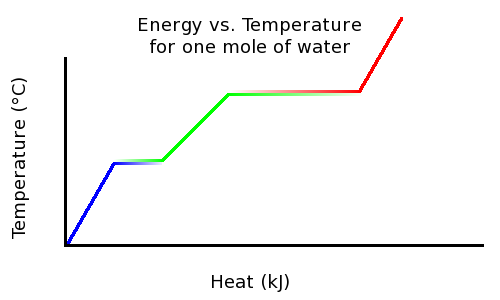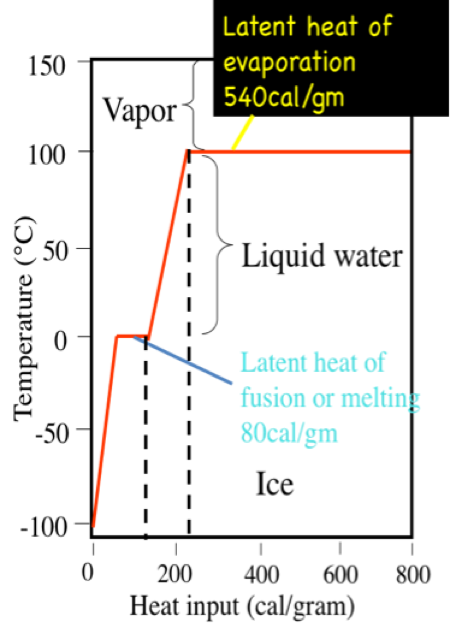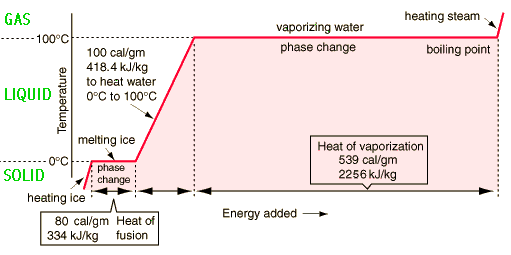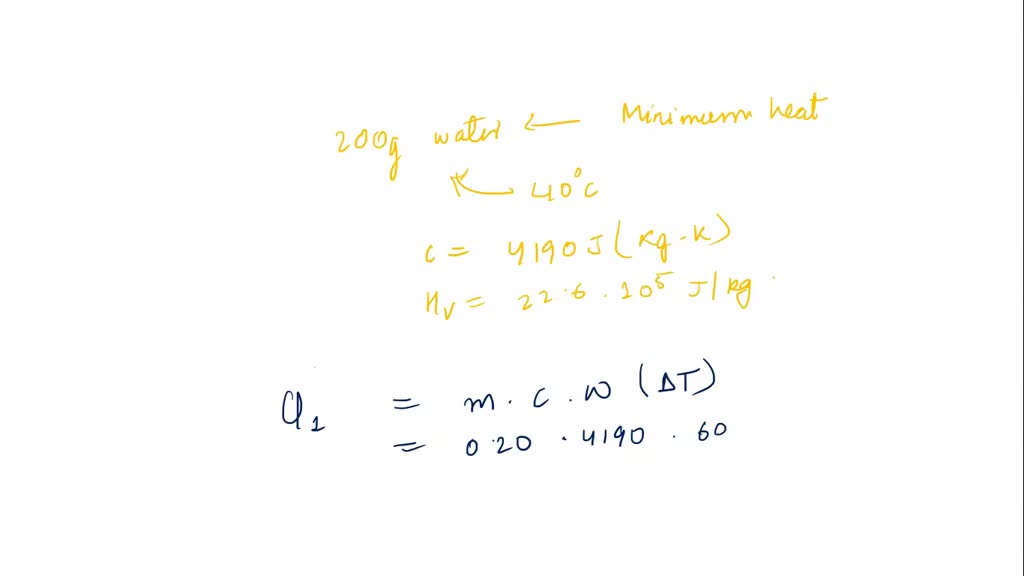
SOLVED: The amount of heat energy needed to heat 200 g of water from 15 °C to its boiling point, and boil it, is

Find how long it takes to bring a cup of water to boiling temperature in a 600 watt microwave 1-28 - YouTube

Find the heat energy required to boil 5 kg of water iff its initial temperature is 30^(@)C (specific heat of water is 4200 J kg^(-1)" "k^(-1) and boiling point is 100^(@)C)

Question Video: Finding the Time a Device Is Used for given the Useful Energy Output, Efficiency, and Total Power Output | Nagwa
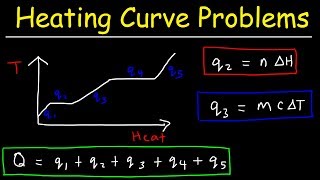
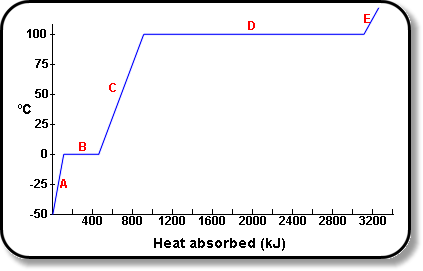
How Much Thermal Energy Is Required To Heat Ice Into Steam - Heating Curve Chemistry Problems - YouTube

Question Video: Finding the Amount of Energy Needed to Change the State of Water from Liquid to Gas | Nagwa