
Magainin 2-PGLa Interactions in Membranes - Two Peptides that Exhibit Synergistic Enhancement of Antimicrobial Activity | Bentham Science

The peptide design based on the essential fragment of magainin 2 (Mag2). | Download Scientific Diagram

Effect of membrane potential on pore formation by the antimicrobial peptide magainin 2 in lipid bilayers - ScienceDirect
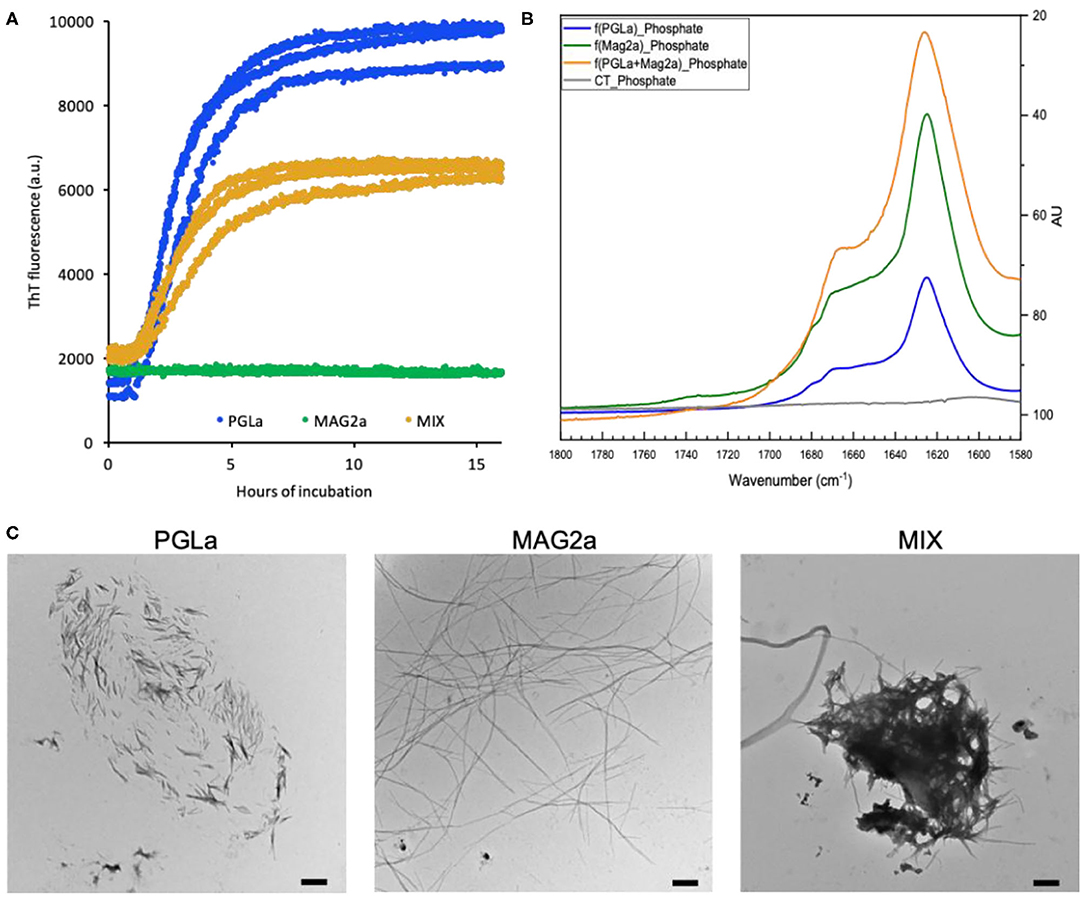
Frontiers | The Reversible Non-covalent Aggregation Into Fibers of PGLa and Magainin 2 Preserves Their Antimicrobial Activity and Synergism

Difference between Magainin-2 and Melittin Assemblies in Phosphatidylcholine Bilayers: Results from Coarse-Grained Simulations | The Journal of Physical Chemistry B
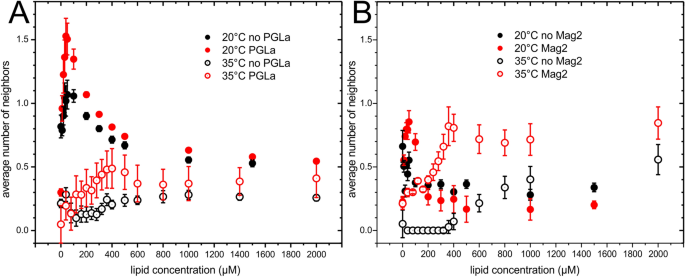
Highly synergistic antimicrobial activity of magainin 2 and PGLa peptides is rooted in the formation of supramolecular complexes with lipids | Scientific Reports
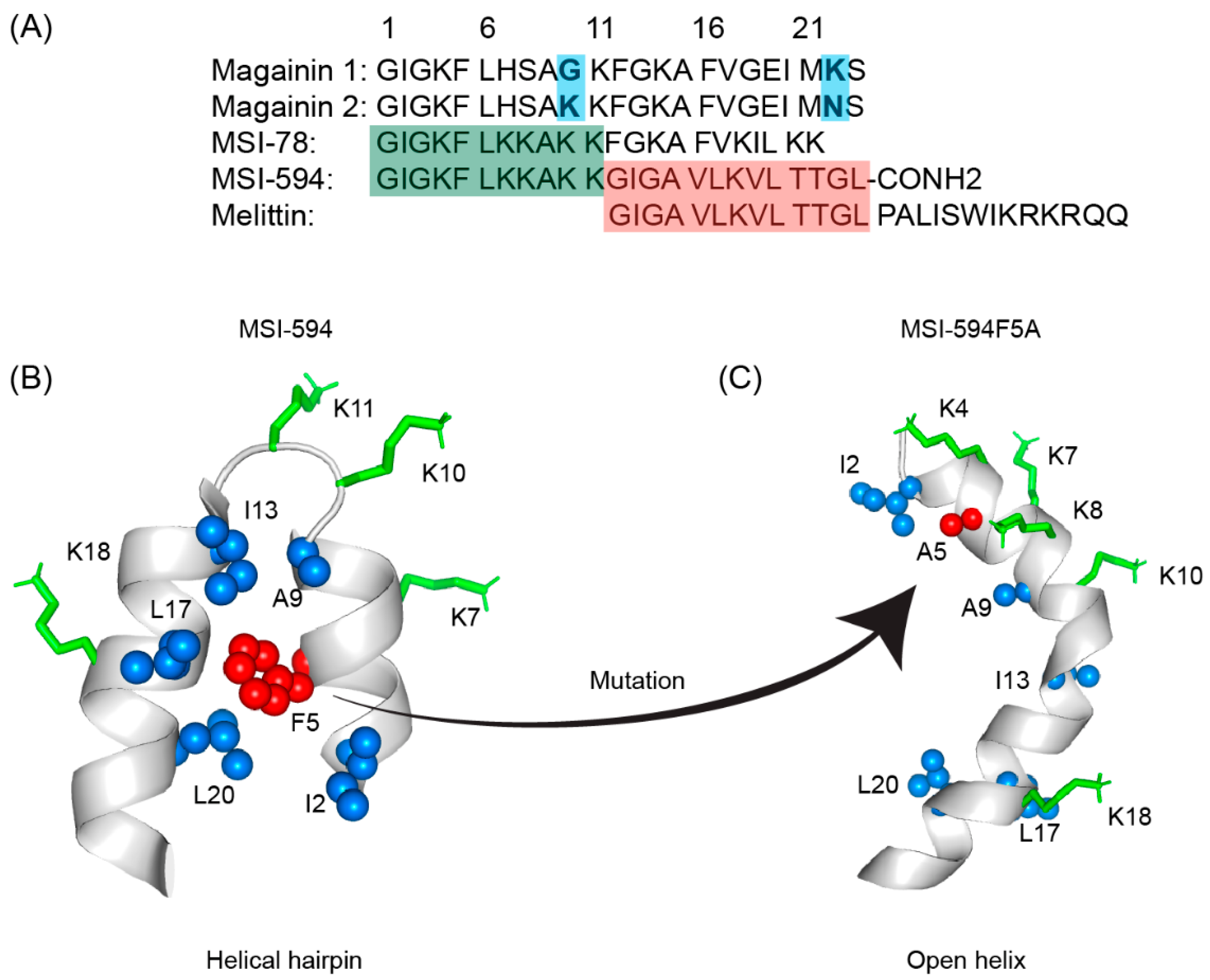
IJMS | Free Full-Text | Atomic-Resolution Structures and Mode of Action of Clinically Relevant Antimicrobial Peptides

Interaction between Antimicrobial Peptide Magainin 2 and Nonlipid Components in the Bacterial Outer Envelope | The Journal of Physical Chemistry B

Comparison of the Membrane Association of Two Antimicrobial Peptides, Magainin 2 and Indolicidin: Biophysical Journal

Structure of Magainin and Alamethicin in Model Membranes Studied by X-Ray Reflectivity: Biophysical Journal

Primary sequence of magainin-2 with side and end-on repre- sentations... | Download Scientific Diagram
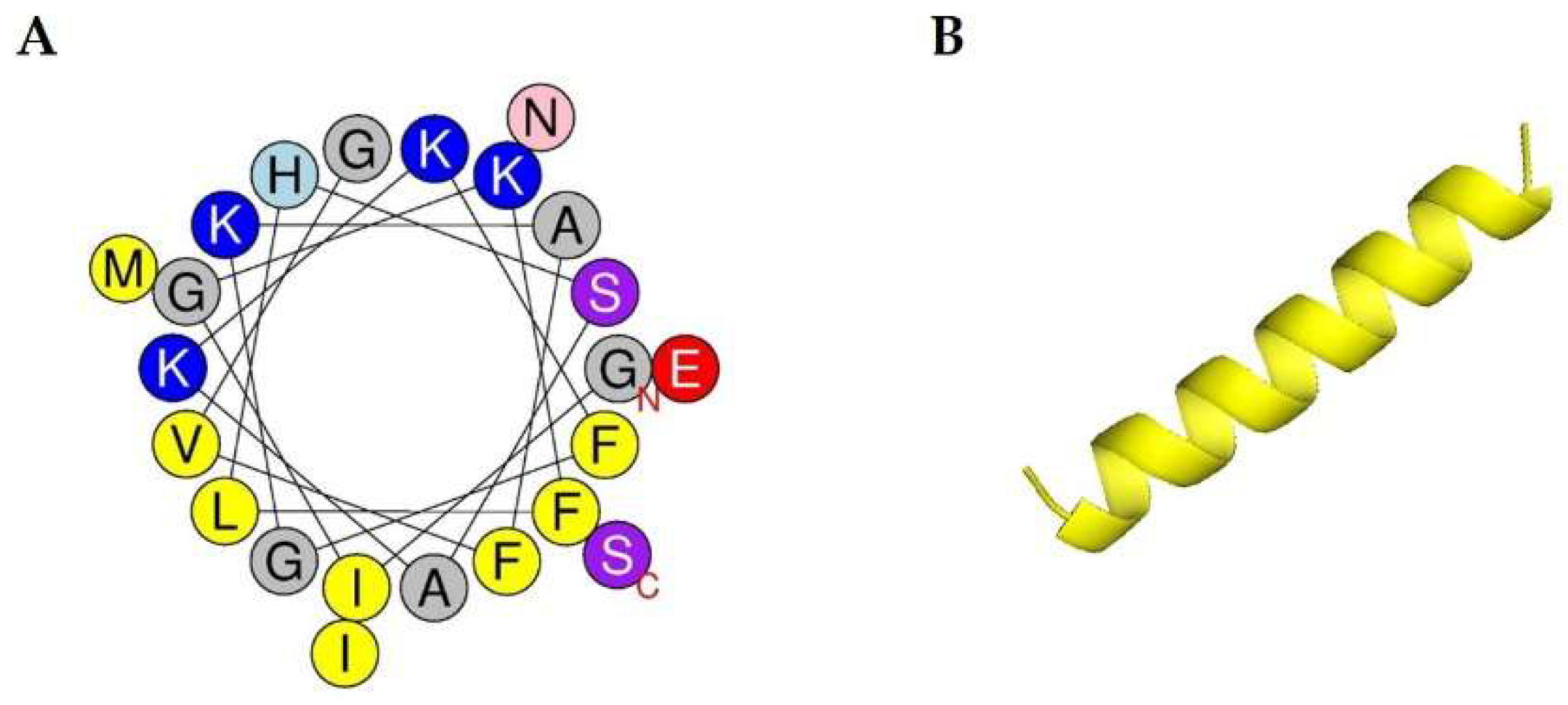
IJMS | Free Full-Text | Antibacterial and Antibiofilm Activity and Mode of Action of Magainin 2 against Drug-Resistant Acinetobacter baumannii
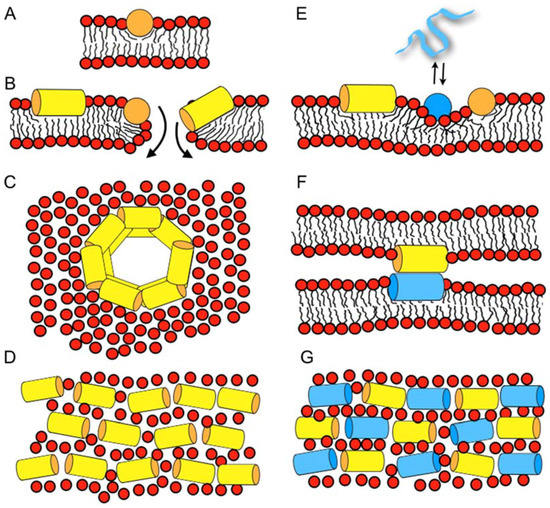
Biomolecules | Free Full-Text | Biophysical Investigations Elucidating the Mechanisms of Action of Antimicrobial Peptides and Their Synergism
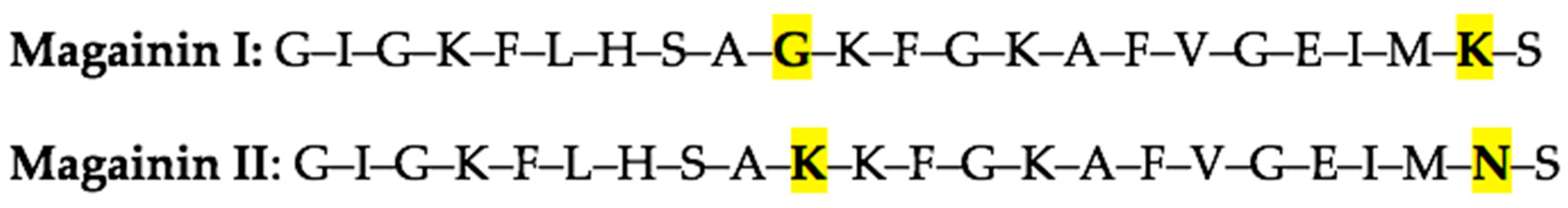
Molecules | Free Full-Text | Review: Examining the Natural Role of Amphibian Antimicrobial Peptide Magainin





![PDF] Helical peptoid mimics of magainin-2 amide. | Semantic Scholar PDF] Helical peptoid mimics of magainin-2 amide. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/72b61153941e4479af51f8d58ba2ad832ae8f7e5/1-Table1-1.png)