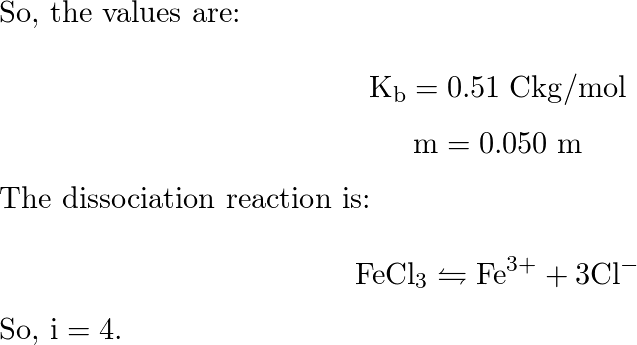Arrange the following in order of increasing boiling point MgCl2 He H2O CH3Cl Ar CO2 I know the cor - YouTube
Aqueous solutions in the order of their increasing boiling points 1) 0.0001 M NaCl 2)0.001 M MgCl2 3)0.001 M urea 4) 0.001 M Nacl
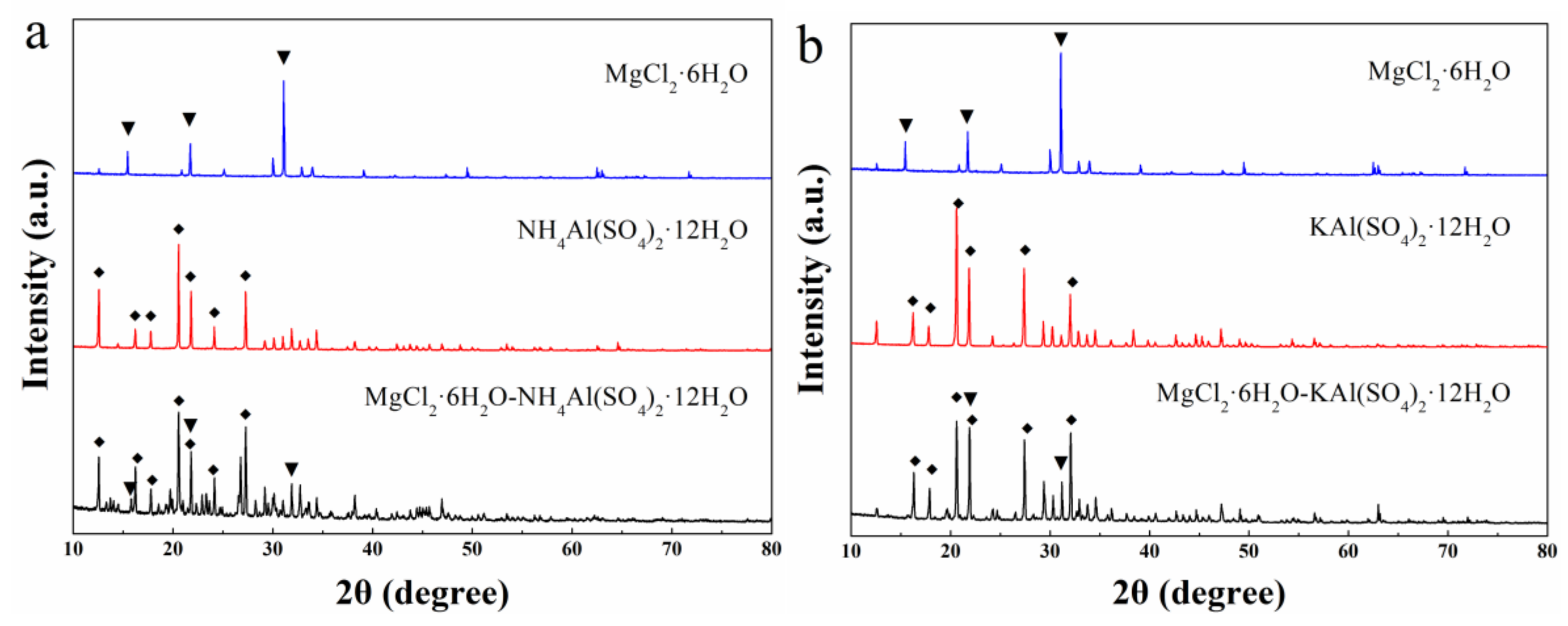
Molecules | Free Full-Text | Compounding MgCl2·6H2O with NH4Al(SO4)2·12H2O or KAl(SO4)2·12H2O to Obtain Binary Hydrated Salts as High-Performance Phase Change Materials

Question Video: Understanding the Difference in Boiling and Freezing Points between Magnesium Chloride and Sodium Chloride Solutions of the Same Concentration | Nagwa

Colligative property measurement is one of the techniques used in the measurement of chemical quantities with reasonable accuracy.If a 40.65 gm sample K2SO4 and BaSO4 is dissolved in 900 gm of pure

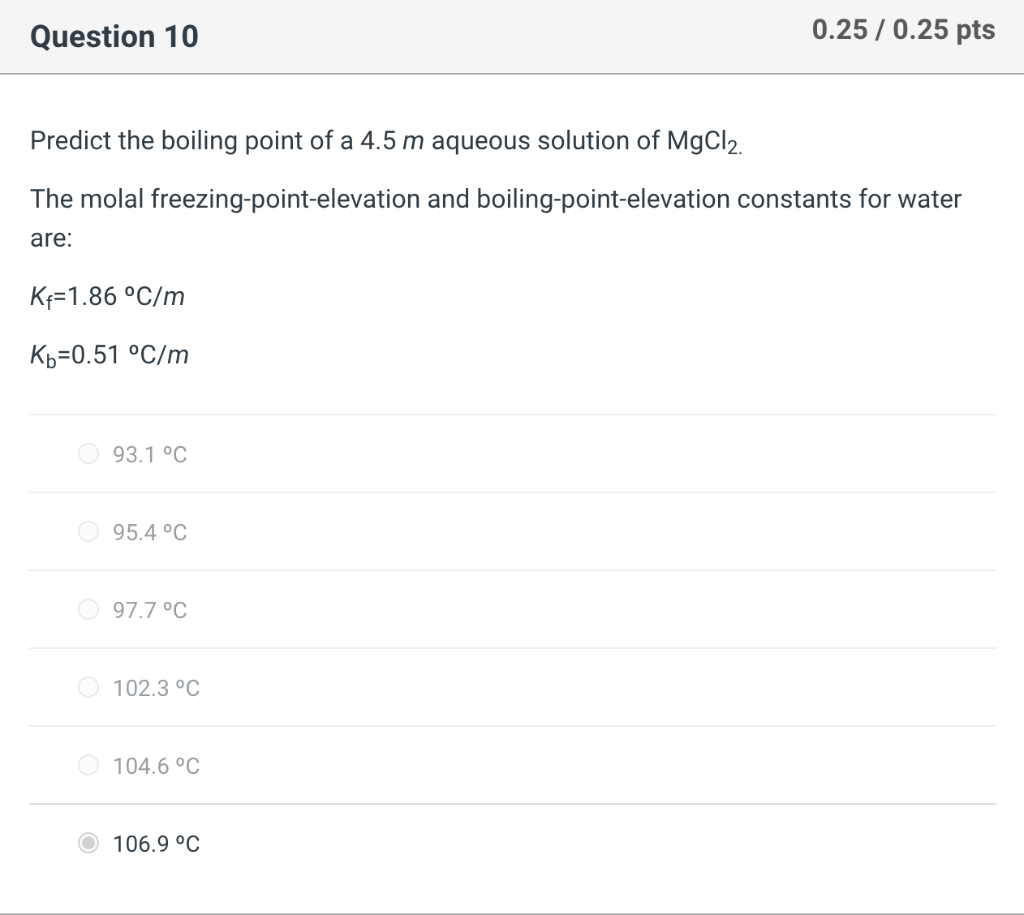
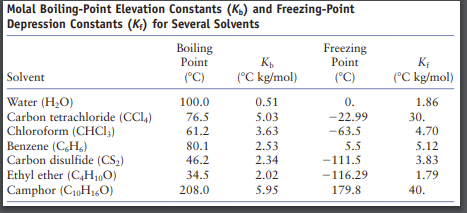
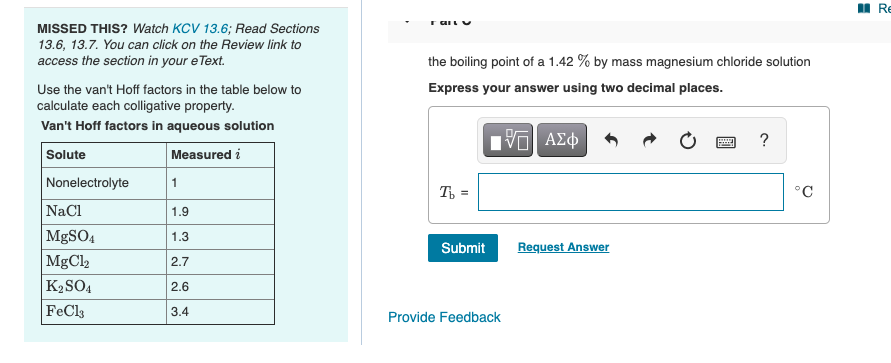
OneClass: Calculate the freezing point and boiling point of each of the following solutions. a. 0.050...

The elevation in boiling point of a solution of 9.43g of MgCl2 in 1kg of water is : ( Kb = 0.52K kg mol^-1 , Molar mass of MgCl2 = 94.3g mol^-1 )
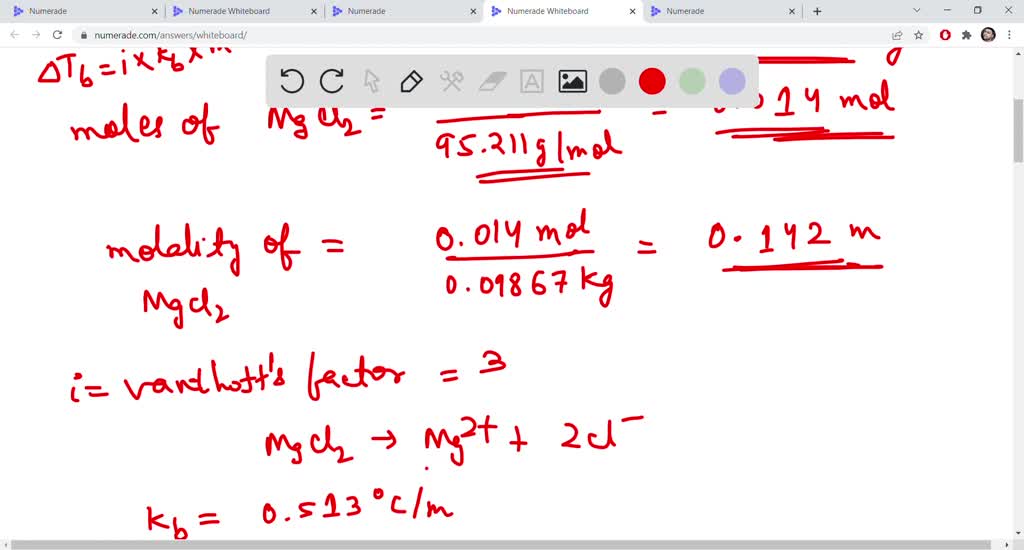
SOLVED: the boiling point ofa 1.33 % by mass magnesium chloride solution Express your answer using two decimal places:

a) Calculate the freezing point of the solution when 1.9 g of MgCl2 (M = 95 g mol−1) was dissolved in 50 g of water, assuming MgCl2 undergoes complete ionization. (Kf for

Comparison of experimental and calculated osmotic coefficients of MgCl2... | Download Scientific Diagram